FULL PRESCRIBING INFORMATION
WARNING: RISK OF THYROID C-CELL TUMORS
- In products with glucagon-like peptide-1 (GLP-1) receptor agonist activity that are pharmacologically active in rats and mice, rodent thyroid C-cell tumors (adenomas and carcinomas) have been observed at clinically relevant exposures and are considered GLP-1 receptor-dependent effects in rodents. Orforglipron is not pharmacologically active in rats or mice and did not produce tumors in rodents [see Nonclinical Toxicology (13.1)]. While orforglipron is pharmacologically active at the human GLP-1 receptor, the human relevance of GLP-1 receptor-dependent thyroid C-cell tumors observed in rodents has not been determined [see Warnings and Precautions (5.1), Nonclinical Toxicology (13.1)].
- FOUNDAYO is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) [see Contraindications (4)]. Counsel patients regarding the potential risk for MTC with the use of FOUNDAYO and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with FOUNDAYO [see Contraindications (4), Warnings and Precautions (5.1)].
1 INDICATIONS AND USAGE
FOUNDAYOTM is indicated in combination with a reduced-calorie diet and increased physical activity to reduce excess body weight and maintain weight reduction long term in adults with obesity or adults with overweight in the presence of at least one weight-related comorbid condition.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Administration
Recommended Administration
- Take FOUNDAYO orally once daily, with or without food.
- Swallow tablets whole. Do not break, crush, or chew.
- Do not take more than one tablet per day.
Recommended Dosage Escalation
Follow the FOUNDAYO starting dosage and escalation described below to reduce the risk of gastrointestinal (GI) adverse reactions [see Warnings and Precautions (5.3), Adverse Reactions (6)].
- The starting dosage is 0.8 mg orally once daily. After at least 30 days on the 0.8 mg dosage, increase the dosage to 2.5 mg once daily.
- After at least 30 days on the 2.5 mg dosage, increase the dosage to 5.5 mg once daily.
- The dosage may be increased to the next dosage level (9 mg, 14.5 mg, or 17.2 mg once daily) after at least 30 days on the current dosage, based on treatment response and tolerability.
- The maximum dosage of FOUNDAYO is 17.2 mg once daily.
2.2 Dosage Modification for Concomitant Use with CYP3A4 Inhibitors and CYP3A4 Inducers
FOUNDAYO dosage modification may be required to manage interactions with some concomitant medications [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Strong CYP3A4 Inhibitors
- Avoid strong CYP3A4 inhibitors that also inhibit OATP1B when taking FOUNDAYO.
- The maximum dosage of FOUNDAYO is 9 mg once daily when used concomitantly with a strong CYP3A4 inhibitor [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
Strong and Moderate CYP3A4 Inducers
2.3 Recommendations Regarding Missed Dose
- If a dose is missed, instruct patients to take the dose as soon as possible.
- Advise patients not to double up the next dose.
- If 7 or more consecutive doses are missed, reinitiate dosage escalation at a lower dosage to reduce the risk of gastrointestinal adverse reactions [see Dosage and Administration (2.1), Warnings and Precautions (5.3), Adverse Reactions (6)].
3 DOSAGE FORMS AND STRENGTHS
FOUNDAYO tablets are available as orforglipron:
- 0.8 mg: pink round tablet debossed with “L” on one side and “G1” on the other side
- 2.5 mg: light yellow round tablet debossed with “L” on one side and “G2” on the other side
- 5.5 mg: grayish purple round tablet debossed with “L” on one side and “G3” on the other side
- 9 mg: pink modified oval tablet debossed with “Lilly” on one side and “G4” on the other side
- 14.5 mg: light yellow modified oval tablet debossed with “Lilly” on one side and “G5” on the other side
- 17.2 mg: grayish purple modified oval tablet debossed with “Lilly” on one side and “G6” on the other side
4 CONTRAINDICATIONS
FOUNDAYO is contraindicated in patients with:
- A personal or family history of MTC or in patients with MEN 2 [see Warnings and Precautions (5.1)].
- Known serious hypersensitivity to orforglipron or any of the excipients in FOUNDAYO. Serious hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with GLP-1 receptor agonists [see Warnings and Precautions (5.6), Adverse Reactions (6.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Thyroid C-Cell Tumors
In products with GLP-1 receptor agonist activity that are pharmacologically active in rats and mice, rodent thyroid C-cell tumors (adenomas and carcinomas) have been observed at clinically relevant exposures and are considered GLP-1 receptor-dependent effects in rodents. Orforglipron is not pharmacologically active in rats or mice and did not produce tumors in rodents [see Nonclinical Toxicology (13.1)]. While orforglipron is pharmacologically active at the human GLP-1 receptor, the human relevance of GLP-1 receptor-dependent thyroid C-cell tumors observed in rodents has not been determined [see Nonclinical Toxicology (13.1)].
Cases of MTC in patients treated with liraglutide, another GLP-1 receptor agonist, have been reported in the postmarketing period; the data in these reports are insufficient to establish or exclude a causal relationship between MTC and GLP-1 receptor agonist use in humans.
FOUNDAYO is contraindicated in patients with a personal or family history of MTC or in patients with MEN 2. Counsel patients regarding the potential risk for MTC with the use of FOUNDAYO and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, or persistent hoarseness).
Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with FOUNDAYO. Such monitoring may increase the risk of unnecessary procedures, due to the low test specificity for serum calcitonin and a high background incidence of thyroid disease. Significantly elevated serum calcitonin values may indicate MTC and patients with MTC usually have calcitonin values >50 ng/L. If serum calcitonin is measured and found to be elevated, the patient should be further evaluated. Patients with thyroid nodules noted on physical examination or neck imaging should also be further evaluated.
5.2 Acute Pancreatitis
Acute pancreatitis has been reported in patients treated with FOUNDAYO. Fatal and non-fatal hemorrhagic or necrotizing pancreatitis have been observed in patients treated with GLP-1 receptor agonists [see Adverse Reactions (6)]. After initiation of FOUNDAYO, observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back) and which may or may not be accompanied by nausea or vomiting. If pancreatitis is suspected, discontinue FOUNDAYO and initiate appropriate management.
5.3 Severe Gastrointestinal Reactions
Use of FOUNDAYO has been associated with gastrointestinal adverse reactions, sometimes severe. In clinical trials, severe gastrointestinal adverse reactions were reported more frequently among patients treated with orforglipron (approximately 3%) than patients who received placebo (1%). Severe gastrointestinal adverse reactions have also been reported postmarketing with GLP-1 receptor agonists [see Adverse Reactions (6)]. FOUNDAYO is not recommended in patients with severe gastroparesis.
5.4 Acute Kidney Injury Due to Volume Depletion
There have been reports of acute kidney injury, in some cases requiring hemodialysis, in patients treated with GLP-1 receptor agonists or FOUNDAYO. The majority of the reported events occurred in patients who experienced gastrointestinal adverse reactions leading to dehydration such as nausea, vomiting, or diarrhea [see Adverse Reactions (6)]. Monitor renal function in patients reporting adverse reactions to FOUNDAYO that could lead to volume depletion, especially during dosage initiation and escalation of FOUNDAYO.
5.5 Hypoglycemia
FOUNDAYO lowers blood glucose and can cause hypoglycemia.
In a trial of adults with type 2 diabetes and BMI ≥27 kg/m2 (Trial 2), hypoglycemia (plasma glucose <54 mg/dL) was reported in 2% of patients treated with orforglipron versus 0.2% of patients receiving placebo. One patient treated with orforglipron and no patients receiving placebo reported severe hypoglycemia in Trial 2. In Trial 2, 7% of patients treated with orforglipron once daily in combination with sulfonylurea reported hypoglycemia compared with 0.5% of patients not taking a sulfonylurea [see Adverse Reactions (6.1)]. There is also increased risk of hypoglycemia in patients treated with FOUNDAYO in combination with insulin [see Drug Interactions (7.2)]. Hypoglycemia has also been associated with FOUNDAYO and GLP-1 receptor agonists in adults without type 2 diabetes [see Adverse Reactions (6.1)].
Inform patients of the risk of hypoglycemia and educate them on the signs and symptoms of hypoglycemia. In patients with diabetes, monitor blood glucose prior to starting FOUNDAYO and during FOUNDAYO treatment. The risk of hypoglycemia may be lowered by a reduction in the dose of insulin or sulfonylurea (or other concomitantly administered insulin secretagogue).
5.6 Hypersensitivity Reactions
Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema) have been reported with GLP-1 receptor agonists [see Adverse Reactions (6.2)]. If hypersensitivity reactions occur, advise the patient to promptly seek medical attention and discontinue use of FOUNDAYO. FOUNDAYO is contraindicated in patients with a prior serious hypersensitivity reaction to orforglipron or to any of the excipients in FOUNDAYO. Use caution in a patient with a history of anaphylaxis or angioedema with another GLP-1 receptor agonist because it is unknown whether such patients will be predisposed to these reactions with FOUNDAYO.
5.7 Diabetic Retinopathy Complications in Patients with Type 2 Diabetes
Temporary worsening of diabetic retinopathy has been reported with rapid improvement in glucose control. FOUNDAYO has not been studied in patients with diabetic retinopathy and/or macular edema requiring acute treatment. Monitor patients with a history of diabetic retinopathy for progression of diabetic retinopathy.
5.8 Acute Gallbladder Disease
Treatment with FOUNDAYO and GLP-1 receptor agonists is associated with an increased occurrence of acute gallbladder disease. In a pool of two clinical trials for weight reduction (Trials 1 and 2), cholelithiasis was reported in 1% of patients treated with orforglipron once daily and 0.7% of placebo-treated patients, and acute cholecystitis was reported in 0.4% of patients treated with orforglipron once daily and 0.3% of placebo-treated patients [see Adverse Reactions (6)]. Acute gallbladder events were associated with weight reduction. If cholecystitis is suspected, gallbladder diagnostic studies and appropriate clinical follow-up are indicated.
5.9 Pulmonary Aspiration During General Anesthesia or Deep Sedation
FOUNDAYO delays gastric emptying [see Clinical Pharmacology (12.2)]. There have been rare postmarketing reports of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation who had residual gastric contents despite reported adherence to preoperative fasting recommendations.
Available data are insufficient to inform recommendations to mitigate the risk of pulmonary aspiration during general anesthesia or deep sedation in patients taking FOUNDAYO, including whether modifying preoperative fasting recommendations or temporarily discontinuing FOUNDAYO could reduce the incidence of retained gastric contents. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking FOUNDAYO.
6 ADVERSE REACTIONS
The following serious adverse reactions are described below or elsewhere in the prescribing information:
- Risk of Thyroid C-Cell Tumors [see Warnings and Precautions (5.1)]
- Acute Pancreatitis [see Warnings and Precautions (5.2)]
- Severe Gastrointestinal Reactions [see Warnings and Precautions (5.3)]
- Acute Kidney Injury Due to Volume Depletion [see Warnings and Precautions (5.4)]
- Hypoglycemia [see Warnings and Precautions (5.5)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.6)]
- Diabetic Retinopathy Complications in Patients with Type 2 Diabetes [see Warnings and Precautions (5.7)]
- Acute Gallbladder Disease [see Warnings and Precautions (5.8)]
- Pulmonary Aspiration During General Anesthesia or Deep Sedation [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of FOUNDAYO has been established in combination with a reduced-calorie diet and increased physical activity to reduce excess body weight and maintain weight reduction long term in adults with obesity or adults with overweight in the presence of at least one weight-related comorbid condition based on adequate and well-controlled trials of an investigational orforglipron formulation (Trials 1 and 2), referred to in this section as FOUNDAYO [see Clinical Studies (14)]. This section of labeling presents safety data from administration of the investigational orforglipron formulation shown as equivalent dosages of once daily FOUNDAYO [see Dosage and Administration (2.1)].
Adverse Reactions in Patients in Weight Management Clinical Trials
Pool of Two Placebo-Controlled Clinical Trials: FOUNDAYO was evaluated for safety in a pool of two randomized, double-blind, placebo-controlled trials that included 3155 adult patients with obesity or overweight treated with FOUNDAYO once daily for up to 72 weeks and a 2-week off-drug follow-up period (Trial 1 and Trial 2) [see Clinical Studies (14)]. The mean age of patients was 49 years and 41% were male. The population was 60% White, 25% Asian, 8% Black or African American, and 0.3% American Indian or Alaska Native; 35% identified as Hispanic or Latino ethnicity. At baseline, patients had an average BMI of 36.5 kg/m2, 51% with a BMI ≥35 kg/m2, 50% with hypertension, 49% with dyslipidemia, 31% with type 2 diabetes, 11% with obstructive sleep apnea, 3% with coronary artery disease, and 3% with cerebrovascular disease.
Across both trials, 8% of patients treated with FOUNDAYO (5.5 mg, 6%; 9 mg, 9%; and 17.2 mg, 10%) once daily permanently discontinued treatment as a result of adverse reactions compared to 3% of patients receiving placebo. The majority of patients (5%) who discontinued FOUNDAYO due to adverse reactions did so due to gastrointestinal adverse reactions.
Common Adverse Reactions
Table 1 shows common adverse reactions associated with the use of once daily FOUNDAYO in the pool of two placebo-controlled trials for weight management (Trials 1 and 2). These adverse reactions occurred more commonly with once daily FOUNDAYO than with placebo and occurred in at least 5% of patients treated with FOUNDAYO.
|
a Includes other related terms. |
||||
| Adverse Reaction | Placebo (N=1,576) % |
FOUNDAYO 5.5 mg once daily (N=1,051) % |
FOUNDAYO 9 mg once daily (N=1,055) % |
FOUNDAYO 17.2 mg once daily (N=1,049) % |
| Nausea | 10 | 26 | 34 | 35 |
| Constipation | 9 | 20 | 27 | 24 |
| Diarrhea | 11 | 21 | 23 | 25 |
| Vomiting | 4 | 13 | 21 | 24 |
| Dyspepsia | 4 | 12 | 16 | 13 |
| Abdominal paina | 7 | 13 | 14 | 14 |
| Headache | 7 | 8 | 9 | 9 |
| Abdominal distension | 3 | 7 | 9 | 8 |
| Fatiguea | 4 | 6 | 7 | 9 |
| Eructation | 1 | 6 | 8 | 8 |
| Gastroesophageal reflux disease | 2 | 6 | 6 | 7 |
| Flatulence | 2 | 5 | 6 | 6 |
| Hair lossa | 2 | 4 | 4 | 5 |
Gastrointestinal Adverse Reactions
In a pool of Trials 1 and 2, gastrointestinal adverse reactions occurred more frequently among patients treated with once daily FOUNDAYO 5.5 mg (60%), 9 mg (68%), and 17.2 mg (69%) than placebo (37%). More patients treated with once daily FOUNDAYO 5.5 mg (3%), 9 mg (6%), and 17.2 mg (6%) discontinued treatment due to gastrointestinal adverse reactions than patients receiving placebo (0.7%).
Of the FOUNDAYO-treated patients who reported GI adverse reactions, 60%, 36%, and 4% reported mild or moderate or severe adverse reactions, respectively. The incidence of nausea, vomiting, and diarrhea was higher during the FOUNDAYO dosage escalation period and decreased over time.
Hypoglycemia
In Trial 2, a trial of patients with type 2 diabetes and BMI ≥27 kg/m2, hypoglycemia (glucose <54 mg/dL) was reported in 2% of FOUNDAYO-treated patients versus 0.2% of placebo-treated patients. One patient treated with FOUNDAYO 5.5 mg once daily, and no patients receiving placebo reported severe hypoglycemia in Trial 2. In this trial, 7% of patients taking FOUNDAYO in combination with sulfonylurea reported hypoglycemia compared with 0.5% of patients not taking sulfonylurea.
In Trial 1, a trial of FOUNDAYO in adults with BMI ≥27 kg/m2 without type 2 diabetes, there was no systematic capturing of hypoglycemia, but glucose <54 mg/dL was reported in 0.6% of FOUNDAYO-treated patients and no placebo-treated patients. No patients in Trial 1 reported severe hypoglycemia.
Acute Pancreatitis
In a pool of Trials 1 and 2, 6 events of acute pancreatitis were confirmed by adjudication in 6 FOUNDAYO-treated patients (0.14 patients per 100 years of exposure) versus 2 events in 1 placebo-treated patient (0.04 patients per 100 years of exposure).
Acute Kidney Injury
In a pool of Trials 1 and 2, acute kidney injury was reported in 0.2% of FOUNDAYO-treated patients compared to 0.05% of placebo-treated patients.
Hypotension
In a pool of Trials 1 and 2, hypotension occurred more frequently among patients taking FOUNDAYO (2%) than patients taking placebo (0.5%). Hypotension was more frequently seen in FOUNDAYO-treated patients on concomitant antihypertensive therapy (4%) compared to FOUNDAYO-treated patients not on antihypertensive therapy (1%).
Acute Gallbladder Disease
In a pool of Trials 1 and 2, cholelithiasis was reported in 1% of FOUNDAYO-treated patients and 0.7% of placebo-treated patients, and acute cholecystitis was reported in 0.4% of FOUNDAYO-treated patients and 0.3% of placebo-treated patients.
Tachycardia
In a pool of Trials 1 and 2, tachycardia (tachycardia, heart rate increased, and sinus tachycardia) was reported in 3% of patients treated with FOUNDAYO and 0.9% receiving placebo. Treatment with FOUNDAYO resulted in a mean increase in heart rate of 4 to 5 beats per minute from baseline compared to 0.5 beat per minute with placebo.
Hair Loss
Hair loss adverse reactions in FOUNDAYO-treated patients were associated with weight reduction. In a pool of Trials 1 and 2, hair loss was reported more frequently in female than male patients in the FOUNDAYO (7% female versus 0.9% male) and placebo (3% female versus 0.7% male) treatment groups.
Dizziness
In a pool of Trials 1 and 2, dizziness was reported in 4% of FOUNDAYO-treated patients and 3% of placebo-treated patients.
Dysgeusia
In a pool of Trials 1 and 2, dysgeusia was reported in 0.9% of FOUNDAYO-treated patients and 0.3% of placebo-treated patients.
Laboratory Abnormalities
Amylase and Lipase Increase
In a pool of Trials 1 and 2, treatment with FOUNDAYO resulted in mean increases from baseline in serum pancreatic amylase concentrations of 16% to 20% and serum lipase concentrations of 26% to 31%, compared to mean increases from baseline in serum pancreatic amylase of 3% and serum lipase of 4% in placebo-treated patients. The clinical significance of elevations in amylase or lipase with FOUNDAYO is unknown in the absence of other signs and symptoms of pancreatitis.
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval use of GLP-1 receptor agonists. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: acute pancreatitis, hemorrhagic and necrotizing pancreatitis sometimes resulting in death; ileus, intestinal obstruction, severe constipation including fecal impaction
Hypersensitivity: anaphylaxis, angioedema
Pulmonary: Pulmonary aspiration has occurred in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation
Renal and Urinary Disorders: acute renal failure or worsening of chronic renal failure, sometimes requiring hemodialysis
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on FOUNDAYO
Table 2 includes clinically relevant interactions where concomitant use of other drugs affects FOUNDAYO.
| Strong CYP3A4 Inhibitors | |
| Intervention | The maximum dosage of FOUNDAYO is 9 mg once daily when used concomitantly with a strong CYP3A4 inhibitor. Avoid concomitant use of FOUNDAYO with strong CYP3A4 inhibitors that also inhibit OATP1B (e.g., ritonavir) [see Dosage and Administration (2.2)]. |
| Clinical Impact | CYP3A4 inhibitors increase FOUNDAYO exposure [see Clinical Pharmacology (12.3)], which may increase the risk of FOUNDAYO-associated adverse reactions. Strong CYP3A4 inhibitors that also clinically inhibit OATP1B are expected to significantly increase plasma concentrations of FOUNDAYO, which may increase the risk of FOUNDAYO-associated adverse reactions [see Warnings and Precautions (5.3), Adverse Reactions (6.1)]. |
| Strong CYP3A4 Inducers | |
| Intervention | Avoid concomitant use of FOUNDAYO with strong CYP3A4 inducers. |
| Clinical Impact | Induction of CYP3A4 decreases FOUNDAYO exposure [see Clinical Pharmacology (12.3)], which may reduce the effectiveness of FOUNDAYO. |
| Moderate CYP3A4 Inducers | |
| Intervention | Monitor FOUNDAYO effectiveness and escalate dosage as needed when used concomitantly with moderate CYP3A4 inducers [see Dosage and Administration (2.1)]. |
| Clinical Impact | Induction of CYP3A4 decreases FOUNDAYO exposure [see Clinical Pharmacology (12.3)], which may reduce the effectiveness of FOUNDAYO. |
7.2 Effect of FOUNDAYO on Other Drugs
Table 3 includes clinically relevant interactions where concomitant use of FOUNDAYO affects other drugs.
| Simvastatin | |
| Intervention | Do not exceed simvastatin 20 mg once daily when concomitantly used with FOUNDAYO. |
| Clinical Impact | Use of FOUNDAYO with simvastatin increased exposure of the active metabolite simvastatin acid two-fold [see Clinical Pharmacology (12.3)]. A two-fold increase in simvastatin acid exposure at the highest simvastatin dose could be clinically meaningful. |
| Insulin or Insulin Secretagogue (e.g., Sulfonylurea) | |
| Intervention | When initiating FOUNDAYO, consider reducing the dose of concomitantly administered insulin or insulin secretagogues (e.g., sulfonylureas) [see Warnings and Precautions (5.5)]. |
| Clinical Impact | FOUNDAYO stimulates insulin release in the presence of elevated blood glucose concentrations which could increase the risk for hypoglycemia when used in combination with insulin or insulin secretagogues. |
7.3 Effect of FOUNDAYO on Oral Medications
FOUNDAYO delays gastric emptying and thereby has the potential to impact the absorption of concomitantly administered oral medications [see Clinical Pharmacology (12.3)].
Advise patients using oral hormonal contraceptives to switch to a non-oral contraceptive method or add a barrier method of contraception for 30 days after initiation with FOUNDAYO and for 30 days after each dose escalation. Hormonal contraceptives that are not administered orally should not be affected.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There will be a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to FOUNDAYO (orforglipron) during pregnancy. Pregnant patients exposed to FOUNDAYO and healthcare providers are encouraged to contact Eli Lilly and Company at 1-800-LillyRx (1-800-545-5979).
Risk Summary
Weight loss offers no benefit to a pregnant patient and may cause fetal harm. When a pregnancy is recognized, advise the pregnant patient of the risk to a fetus and discontinue FOUNDAYO.
There are no adequate and well-controlled studies of FOUNDAYO during pregnancy. Based on animal reproduction studies, there may be risks to the fetus from exposure to orforglipron during pregnancy.
Imbalances in malformations have been reported in rats and rabbits at low multiples of clinical exposure with GLP-1 receptor agonists active in those species. Orforglipron is not pharmacologically active in rats and rabbits.
In animal reproduction studies, oral administration of orforglipron to pregnant monkeys during organogenesis at doses lower than the maximum recommended human dose (MRHD) did not result in embryofetal effects. Higher dose levels and exposures could not be evaluated in monkeys due to dose limiting effects on body weight (see Data).
In a rabbit dose-range finding study, oral administration of orforglipron to pregnant rabbits during organogenesis at exposures 14 times the clinical exposure at the MRHD resulted in external malformations and decreases in fetal and placental weights in the absence of maternal toxicity (see Data). In the definitive rabbit study, oral administration of orforglipron to pregnant rabbits during organogenesis at exposures up to 6 times the clinical exposure at the MRHD did not result in embryofetal effects (see Data).
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The estimated background risk of major birth defects and miscarriage for the indicated population is increased when compared to the general population.
Disease-Associated Maternal and/or Embryo/Fetal Risk
Appropriate weight gain relative to pre-pregnancy weight is currently recommended for all pregnant patients, including those with obesity or overweight, due to the obligatory weight gain that occurs in maternal tissues during pregnancy.
Data
Animal Data
Embryofetal development was evaluated in pregnant monkeys administered orforglipron orally during organogenesis (gestation days 20 to 50) at doses of 0.15 and 0.7 mg/kg/day resulting in systemic exposures lower than the human exposure at the MRHD of 17.2 mg FOUNDAYO, based on AUC. No effects on pregnancy outcome, placental weight, fetal viability, fetal body weight, fetal measurements or external, visceral or skeletal evaluations were observed. Exposure in monkeys was lower than exposure at the MRHD. Higher dose levels and exposures cannot be achieved in pregnant monkeys due to on-target body weight effects.
Orforglipron is not pharmacologically active in rabbits. In a rabbit dose-range finding study, orforglipron was administered orally once daily during organogenesis (gestation days 7 to 19) at dose levels of 2, 20, and 200 mg/kg/day. These doses resulted in exposures that were approximately 0.03, 0.8 and 14 times, respectively, the clinical exposure at the MRHD, based on AUC. Statistically significant decreases in fetal and placental weights and external malformations in 2 fetuses from 2 dams were observed at 200 mg/kg/day. In the definitive rabbit study, orforglipron was given orally once daily during organogenesis (gestation days 7 to 19) at dose levels of 4, 40, and 200 mg/kg/day resulting in exposures approximately 0.06, 1.4, and 6.2 times the clinical exposure at the MRHD, respectively, based on AUC. No embryofetal effects were observed in the definitive rabbit study at any dose level.
Orforglipron is not pharmacologically active in rats. In a pre- and post-natal study in rats, orforglipron was administered orally once daily from implantation through lactation at doses of 5, 30, and 200 mg/kg/day, which resulted in exposures that were approximately 2.9, 4.5, and 13 times the clinical exposure at the MRHD, respectively, based on AUC. No effects were noted at any dose level in the F0 maternal rats or the F1 pups in the pre- and post-natal development study in rats.
In a tissue distribution study, [14C]orforglipron-derived radioactivity was not distributed to fetal tissues in pregnant rats.
8.2 Lactation
Risk Summary
There are no data on the presence of orforglipron or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. Orforglipron was present in the milk of lactating rats in in vivo testing (see Data). When a drug is present in animal milk, it is likely that the drug will be present in human milk. FOUNDAYO is not recommended for nursing women. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for FOUNDAYO and any potential adverse effects on the breastfed child from FOUNDAYO or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Contraception
Based on animal reproduction studies, there may be risks to the fetus from exposure to orforglipron during pregnancy [see Pregnancy (8.1)]. Advise women of childbearing potential to use effective contraception during treatment with FOUNDAYO. The effect of FOUNDAYO on the absorption of oral contraceptives has not been evaluated in a clinical trial. Because delayed gastric emptying may affect the absorption of oral medications, advise patients using oral hormonal contraceptives to switch to a non-oral contraceptive method, or add a barrier method of contraception for 30 days after initiation with FOUNDAYO and for 30 days after each dose escalation [see Drug Interactions (7.3), Clinical Pharmacology (12.3)].
8.4 Pediatric Use
Safety and effectiveness of FOUNDAYO have not been established in pediatric patients.
8.5 Geriatric Use
In a pool of Trials 1 and 2, 399 (13%) FOUNDAYO-treated patients were 65 years of age or older, and 33 (1%) FOUNDAYO-treated patients were 75 years of age or older at baseline [see Clinical Studies (14)]. No overall differences in safety or effectiveness of FOUNDAYO have been observed between patients 65 years of age or older and younger adult patients.
8.6 Hepatic Impairment
FOUNDAYO is not recommended for use in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)].
No dosage modification of FOUNDAYO is recommended in patients with mild or moderate hepatic impairment (Child-Pugh Class A and B, respectively) [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No dosage modification of FOUNDAYO is recommended for patients with renal impairment. In subjects with renal impairment including end-stage renal disease (ESRD), no change in orforglipron pharmacokinetics (PK) was observed [see Clinical Pharmacology (12.3)]. Monitor renal function in patients reporting adverse reactions to FOUNDAYO that could lead to volume depletion [see Warnings and Precautions (5.4)].
10 OVERDOSAGE
In the event of an overdose, consider contacting Poison Control (1-800-222-1222) or a medical toxicologist for latest recommendations. Initiate appropriate supportive treatment according to the patient's clinical signs and symptoms. A period of observation and treatment for these symptoms may be necessary, considering the half-life of orforglipron [see Clinical Pharmacology (12.3)].
11 DESCRIPTION
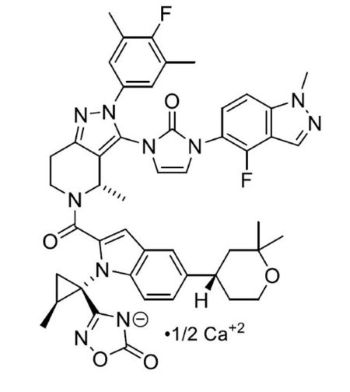
FOUNDAYO contains orforglipron, a GLP-1 receptor agonist. The molecular formula for orforglipron calcium is C48H47F2N10O5.[0.5]Ca and the molecular weight is 902.0 g/mol. The chemical name is calcium bis{3-[(1S,2S)-1-({2-(4-fluoro-3,5-dimethylphenyl)-3-({3-[3-(4-fluoro-1-methyl-1H-indazol-5-yl)-2-oxo-2,3-dihydro-1H-imidazol-1-yl]-4-methyl-2,4,6,7-tetrahydro-5H-pyrazolo[4,3-c]pyridine-5-yl}carbonyl)-5-[(4S)-2,2-dimethyloxan-4-yl]-1H-indol-1-yl}-2-methylcyclopropyl]-5-oxo-1,2,4-oxadiazol-4(5H)-ide}. Orforglipron calcium has the following chemical structure:
Orforglipron calcium is a white to practically white to light brown solid that is hygroscopic. Orforglipron calcium is insoluble in water.
FOUNDAYO tablets contain 0.8 mg, 2.5 mg, 5.5 mg, 9 mg, 14.5 mg, or 17.2 mg of orforglipron (equivalent to 0.817 mg, 2.554 mg, 5.619 mg, 9.194 mg, 14.81 mg, and 17.57 mg of orforglipron calcium, respectively) as film-coated, debossed tablets for oral use. Each tablet contains the following inactive ingredients: copovidone, crospovidone, magnesium stearate, microcrystalline cellulose, and sodium carbonate anhydrous. The tablet film-coating material contains polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. Additionally, the film coating of the:
- 0.8 mg and 9 mg tablets contain ferric oxide red and ferric oxide yellow
- 2.5 mg and 14.5 mg tablets contain ferric oxide yellow
- 5.5 mg and 17.2 mg tablets contain ferrosoferric oxide and ferric oxide red.
Each tablet contains 10 mg or less of sodium.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
FOUNDAYO is a GLP-1 receptor agonist that binds to and activates the human GLP-1 receptor.
GLP-1 is a physiological regulator of appetite and caloric intake. GLP-1 receptors are present in brain regions that regulate appetite. In animal studies, orforglipron distributed to and activated neurons in brain regions that regulate appetite and food intake.
12.2 Pharmacodynamics
FOUNDAYO reduces body weight, with greater fat mass loss than lean mass loss. FOUNDAYO decreases food intake. This effect is likely mediated by decreased appetite.
FOUNDAYO delays gastric emptying. The delay is largest after the first dose and diminishes over time.
12.3 Pharmacokinetics
The pharmacokinetics of orforglipron is similar between healthy subjects and patients with overweight (BMI ≥27 kg/m2) or obesity. Steady state exposure is achieved after approximately 1 week of once daily administration.
The data below refer to FOUNDAYO dosages [see Dosage and Administration (2.1)]. Except in the food effect study, data were generated with the corresponding orforglipron investigational formulation [see Adverse Reactions (6.1)].
Absorption
Maximum concentration of orforglipron is reached 4 to 8 hours post dose. Orforglipron tablet exposure increases in a dose-proportional manner. The geometric mean absolute bioavailability of orforglipron was 77% after a 0.8 mg dose.
Distribution
The mean steady-state volume of distribution of orforglipron is approximately 285 L, following intravenous dosing in healthy subjects. Orforglipron plasma protein binding is greater than 99%. FOUNDAYO is only for oral use.
Elimination
The mean systemic clearance of orforglipron is 7.15 L/hour. The elimination half-life is approximately 29 to 49 hours after an oral dose.
Special Populations
The intrinsic factors of age (18 to 92 years), sex, race (White, Asian, Black or African American, American Indian, Multiracial, or Hawaiian Pacific Islander), ethnicity (Hispanic, Non-Hispanic), body weight (56.4 to 227 kg), and renal impairment (eGFR 27.8 to 156 mL/min/1.73 m2) do not have a clinically relevant effect on the pharmacokinetics of orforglipron.
Patients with Hepatic Impairment
The pharmacokinetics of orforglipron after a single oral 0.8 mg dose was evaluated in patients with mild, moderate, and severe hepatic impairment (Child-Pugh Class A, B, and C, respectively) and in subjects with normal hepatic function. Orforglipron exposure was similar in patients with mild hepatic impairment and normal hepatic function. Orforglipron AUC(0-∞) increased by 1.7-fold and 4.6-fold in patients with moderate and severe hepatic impairment, respectively, compared to subjects with normal hepatic function. Cmax in patients with moderate and severe hepatic impairment was similar to Cmax in subjects with normal hepatic function [see Use in Specific Populations (8.6)].
Patients with Renal Impairment
Renal impairment does not impact the pharmacokinetics of orforglipron. The pharmacokinetics of orforglipron after a single oral 0.8 mg dose were evaluated in patients with severe renal impairment and ESRD compared with subjects who had normal renal function. Data from clinical trials have also shown that renal impairment in patients with overweight or obesity does not impact the pharmacokinetics of orforglipron [see Use in Specific Populations (8.7)].
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Effects of Other Drugs on FOUNDAYO
Table 4 outlines the results of definitive clinical trials and physiologically based pharmacokinetic (PBPK) simulations assessing the effect of other drugs on orforglipron pharmacokinetics. Concomitant use of strong CYP3A4 inhibitors significantly increased exposure of orforglipron while concomitant use of moderate or strong CYP3A4 inducers led to reductions in orforglipron exposure [see Dose and Administration (2.2), Drug Interactions (7.1)].
|
a Simulated using physiologically based pharmacokinetic modeling. |
|||
| Concomitant Drug Category | Concomitant Drug (Dose) | Change in Orforglipron | |
| AUC | Cmax | ||
| Strong CYP3A4 inhibitors | Clarithromycin (500 mg twice daily) | ↑3.5-fold | ↑1.9-fold |
| Moderate CYP3A4 inhibitors | Verapamila (80 mg three times daily) | ↑2-fold | ↑1.6-fold |
| Strong CYP3A4 inducers | Carbamazepine (300 mg twice daily) | ↓82% | ↓55% |
| Moderate CYP3A4 inducers | Efavirenza (600 mg once daily) | ↓61% | ↓33% |
| Weak CYP3A4 inducers | Modafinila (200 mg once daily) | ↓16% | ↓7% |
| OATP1B inhibitors | Cyclosporine (200 mg twice daily) | ↑2.6-fold | ↑1.3-fold |
| P-gp inhibitors | Quinidine (200 mg twice daily) | ↓12% | ↓26% |
| Acid reducing agents | Esomeprazole (40 mg once daily) | No effect | No effect |
Effects of FOUNDAYO on Other Drugs
Table 5 outlines the results of clinical assessments that evaluated the effect of orforglipron on other drugs. Concomitant use of orforglipron and simvastatin led to significant increases in the active metabolite simvastatin acid whether orforglipron was co-administered or dosing was staggered by 2 hours [see Drug Interactions (7.2)]. No clinically relevant changes were observed for orforglipron when used with rosuvastatin or atorvastatin.
| Concomitant Drug Category | Concomitant Drug (Dose) |
Orforglipron Dosage |
Change in Concomitant Drug | |
| AUC | Cmax | |||
| Statin | Simvastatin (prodrug) (20 mg) | 17.2 mg once daily | up to ↓16% | up to ↓27% |
| Simvastatin acid (active metabolite) | 17.2 mg once daily | ↑2- to 2.5-fold | ↑2.3- to 2.5-fold | |
| Atorvastatin (40 mg) | 14.5 mg once daily | ↑1.5-fold | ↑0.9-fold | |
| BCRP substrate | Rosuvastatin (20 mg) | 17.2 mg once daily | ↑1.7-fold | ↑1.3-fold |
| CYP3A4 substrates | Midazolam (200 mcg) | 17.2 mg once daily | ↑1.1-fold | No effect |
| P-gp substrates | Digoxin (0.25 mg) | 17.2 mg once daily | ↑1.2-fold | ↑1.2-fold |
| OATP1B substrates | Endogenous OATP1B biomarker coproporphyrin-1 | up to 14.5 mg once daily | No effect | No effect |
Potential Drug Interactions due to Gastric Emptying Delay
Orforglipron delays gastric emptying and has the potential to affect the rate of absorption of other concomitantly administered oral drugs. The gastric emptying delay effect of orforglipron on acetaminophen Cmax was largest after the first dose of FOUNDAYO 0.8 mg with acetaminophen Cmax decreased by 28%. The effect diminished after repeated dosing of FOUNDAYO 17.2 mg.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Orforglipron is not pharmacologically active in rats or mice. In a 2-year rat carcinogenicity study, orforglipron was non-carcinogenic at oral daily doses of 5, 30, and 200 mg/kg/day resulting in exposures approximately 3, 9, and 26 times the clinical exposure at the MRHD, respectively, based on AUC. In a 26-week study in Tg.RasH2 transgenic mice, orforglipron was non-carcinogenic at oral daily doses of 5, 30, and 200 mg/kg/day. For other GLP-1 receptor agonists that are pharmacologically active in rats, thyroid C-cell hyperplasia, and thyroid C-cell adenoma and carcinoma have been observed at clinically relevant exposures and are considered an on-target class effect. Orforglipron would be expected to have the same on-target class effect if it was pharmacologically active in rats. The human relevance of these findings is unknown.
Orforglipron was not mutagenic or clastogenic in a bacterial reverse mutation test, an in vitro micronucleus test in human lymphoblastoid cells, or in an in vivo bone marrow micronucleus study in rats.
There were no effects on male and female fertility in rats orally administered orforglipron up to 200 mg/kg/day (19 times and 36 times the clinical exposure at the MRHD, respectively, based on AUC).
14 CLINICAL STUDIES
Overview of Trials 1 and 2
The effectiveness of FOUNDAYO has been established in combination with a reduced-calorie diet and increased physical activity to reduce excess body weight and maintain weight reduction long term in adults with obesity or adults with overweight in the presence of at least one weight-related comorbid condition based on adequate and well-controlled trials of an investigational orforglipron formulation (Trials 1 and 2), referred to in this section as FOUNDAYO. This section of labeling presents efficacy data from administration of the investigational orforglipron formulation shown as equivalent dosages of once daily FOUNDAYO [see Dosage and Administration (2.1)].
FOUNDAYO was studied in two randomized, double-blind, placebo-controlled trials (Trials 1 and 2) in adults aged 18 years and older. In these trials, all patients received standard lifestyle intervention which included instruction on reduced-calorie diet and physical activity counseling (recommended minimum of 150 minutes/week) that began with the first dose of trial medication or placebo and continued throughout the trials.
In Trial 1 patients were randomized in a 3:3:3:4 ratio to FOUNDAYO 5.5 mg once daily, 9 mg once daily, 17.2 mg once daily, or placebo once daily. In Trial 2 patients were randomized in a 1:1:1:2 ratio to FOUNDAYO 5.5 mg once daily, 9 mg once daily, 17.2 mg once daily, or placebo once daily. In Trials 1 and 2, dosages were titrated according to the dosage escalation described in Section 2.1. In these trials, the primary efficacy parameter was mean percent change in body weight after 72 weeks of treatment.
Trial 1 (NCT05869903) was a 72-week trial that enrolled 3,127 adult patients with obesity (BMI ≥30 kg/m2), or with overweight (BMI 27 to <30 kg/m2) and at least one weight-related comorbid condition, such as dyslipidemia, hypertension, obstructive sleep apnea, or cardiovascular disease; patients with type 2 diabetes were excluded. At baseline, mean age was 45 years (range 18 to 88 years), 64% were female, 56% were White, 28% were Asian, 9% were Black or African American, and 0.4% were American Indian/Alaska Native. A total of 38% were Hispanic or Latino ethnicity. Mean baseline body weight was 103.2 kg and mean BMI was 37 kg/m2. At baseline, 40% of patients had hypertension, 39% had dyslipidemia, 11% had obstructive sleep apnea, 1.4% had coronary artery disease, and 1.5% had cerebrovascular disease.
Trial 2 (NCT05872620) was a 72-week trial that enrolled 1,613 adult patients with BMI ≥27 kg/m2 and type 2 diabetes. Patients included in the trial had baseline HbA1c 7% to 10% and were treated with either diet and exercise alone, or any oral anti-hyperglycemic agent except dipeptidyl peptidase-4 (DPP-4) inhibitors or GLP-1 receptor agonists. Patients who were taking injectable glucose-lowering agents, including insulin, GLP-1 receptor agonists, or pramlintide were also excluded. At baseline, mean age was 57 years (range 20 to 92 years), 47% were female, 71% were White, 17% were Asian, 7% were Black or African American, and 0.3% were American Indian/Alaska Native. A total of 30% were Hispanic or Latino ethnicity. Mean baseline body weight was 101.4 kg, mean BMI was 35.6 kg/m2, and mean HbA1c was 8%. At baseline, 74% of patients had hypertension, 71% had dyslipidemia, 13% had obstructive sleep apnea, 7% had coronary artery disease, 5% had cerebrovascular disease, and 11% had diabetic retinopathy.
Results for Trials 1 and 2
The proportions of patients who discontinued trial drug in Trial 1 were 22%, 22%, and 24% for the 5.5 mg, 9 mg, and 17.2 mg once daily FOUNDAYO-treated groups, respectively, and 30% for the placebo-treated group. The proportion of patients who discontinued trial drug in Trial 2 were 19%, 22%, and 20% for the 5.5 mg, 9 mg, and 17.2 mg once daily FOUNDAYO-treated groups, respectively, and 20% for the placebo-treated group.
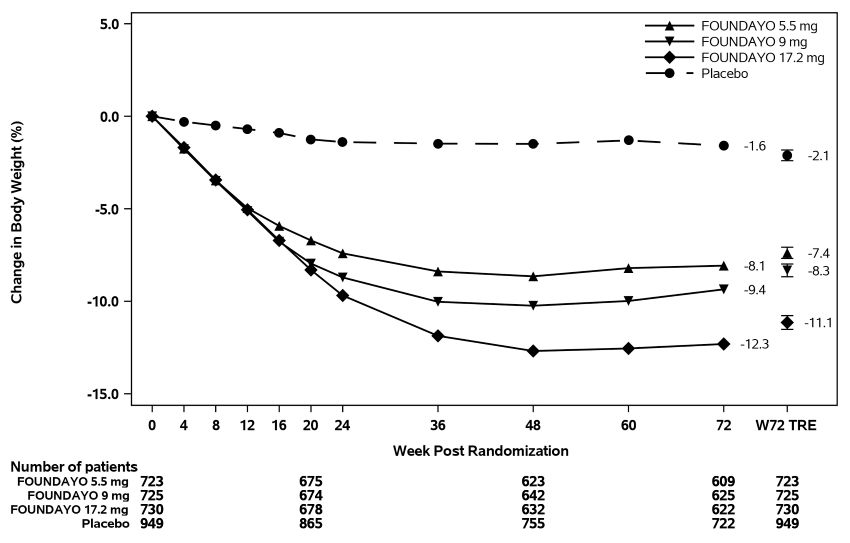
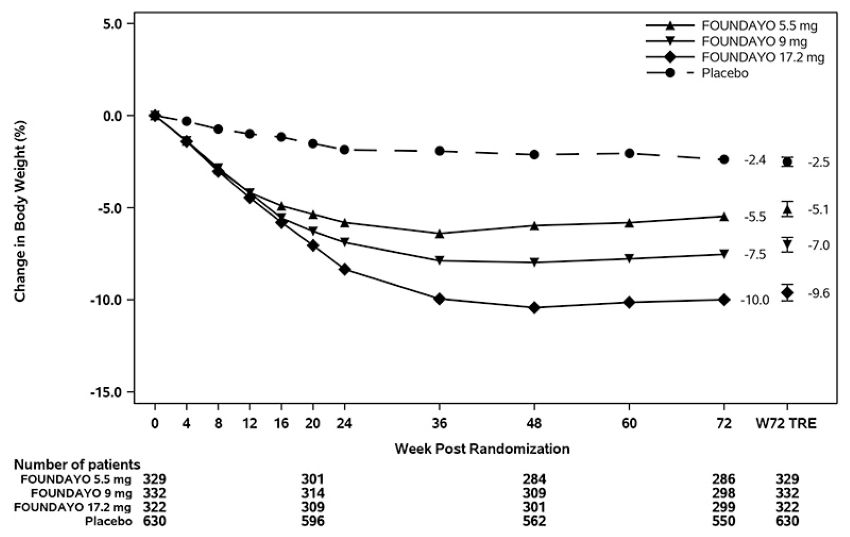
For Trials 1 and 2, the primary efficacy parameter was mean percent change in body weight from baseline to Week 72. After 72 weeks of treatment in Trials 1 and 2, there was a statistically significant reduction in body weight in the FOUNDAYO-treated groups compared with the placebo groups (see Table 6). A reduction in body weight was observed with FOUNDAYO regardless of age, sex, race, ethnicity, baseline BMI, and glycemic status.
|
Abbreviations: ANCOVA = analysis of covariance; CI = confidence interval; N = number of patients randomly assigned to trial drug. Note: Baseline mean is calculated using all randomized patients. |
||||||||
|
a The intent-to-treat population consists of all randomized patients. For Trial 1 at Week 72, the body weight endpoint was missing for 24%, 16%, 14%, and 15% of patients randomized to placebo, FOUNDAYO 5.5 mg once daily, 9 mg once daily and 17.2 mg once daily, respectively. For Trial 2 at Week 72, the body weight endpoint was missing for 13%, 13%, 10%, and 7% of patients randomized to placebo, FOUNDAYO 5.5 mg once daily, 9 mg once daily and 17.2 mg once daily, respectively. Missing data were imputed from retrieved patients of the same randomized treatment group when the missingness was possibly related to trial treatment; otherwise, missing data were imputed using observed data from the same randomized treatment group (primary modified multiple imputation). |
||||||||
|
b Unconditional average treatment effect estimated using ANCOVA adjusted for baseline value and other stratification factors. |
||||||||
|
c p<0.001 (unadjusted 2-sided) compared to placebo for superiority; controlled for multiplicity. |
||||||||
|
d Unconditional average treatment effect estimated using logistic regression adjusted for baseline value and other stratification factors. |
||||||||
|
e Not controlled for multiplicity. |
||||||||
| Trial 1 (without diabetes) | Trial 2 (with type 2 diabetes) | |||||||
| Intent-to-Treat (ITT) Populationa | Placebo once daily N = 949 |
FOUNDAYO 5.5 mg once daily N = 723 |
FOUNDAYO 9 mg once daily N = 725 |
FOUNDAYO 17.2 mg once daily N = 730 |
Placebo once daily N = 630 |
FOUNDAYO 5.5 mg once daily N = 329 |
FOUNDAYO 9 mg once daily N = 332 |
FOUNDAYO 17.2 mg once daily N = 322 |
| Body Weight | ||||||||
| Baseline mean (kg) | 103.9 | 103.2 | 102.2 | 103.1 | 101.2 | 102.3 | 102.7 | 99.8 |
| % Change from baselineb | -2.1 | -7.4 | -8.3 | -11.1 | -2.5 | -5.1 | -7 | -9.6 |
| % difference from placebo (95% CI)b | -5.3 (-6.1, -4.4)c |
-6.2 (-7.1, -5.3)c |
-9 (-10, -8.1)c |
-2.6 (-3.5, -1.6)c |
-4.5 (-5.4, -3.6)c |
-7.1 (-8.1, -6.1)c |
||
| % of patients who lost ≥5% body weight | 26.8 | 59.6 | 63.1 | 71.5 | 26.8 | 47.4 | 54.7 | 67 |
| % difference from placebo (95% CI)d | 32.7 (27.9, 37.6)c |
36.3 (31.5, 41.1)c |
44.7 (40, 49.3)c |
20.6 (13.9, 27.3)c |
27.9 (21.3, 34.5)c |
40.2 (33.8, 46.6)c |
||
| % of patients who lost ≥10% body weight | 13 | 32.5 | 39.8 | 54.5 | 9.2 | 22.5 | 31.1 | 45.6 |
| % difference from placebo (95% CI)d | 19.4 (15.1, 23.7)c |
26.8 (22.4, 31.1)c |
41.4 (37, 45.9)c |
13.3 (8.1, 18.5)c |
21.9 (16.3, 27.5)c |
36.4 (30.5, 42.3)c |
||
| % of patients who lost ≥15% body weight | 6 | 14.3 | 20.1 | 35.9 | 3.1 | 6.7 | 14.4 | 25.9 |
| % difference from placebo (95% CI)d | 8.3 (5.2, 11.4)c |
14.1 (10.7, 17.6)c |
29.8 (25.9, 33.7)c |
3.6 (0.4, 6.8)e |
11.3 (7.3, 15.3)c |
22.8 (17.8, 27.8)c |
||
| % of patients who lost ≥20% body weight | 2.9 | 5.9 | 8.9 | 18.4 | 0.7 | 2.6 | 4.4 | 10.8 |
| % difference from placebo (95% CI)d | 3 (0.9, 5.2)e |
6 (3.5, 8.4)c |
15.5 (12.4, 18.6)c |
1.8 (-0.2, 3.8)e |
3.7 (1.4, 6)e |
10.1 (6.6, 13.5)e |
||
The time courses of weight reduction from baseline with FOUNDAYO and placebo through
Week 72 are depicted in Figure 1 for Trial 1 and Figure 2 for Trial 2.
Figure 1: Change Over Time in Body Weight in Trial 1 in Patients with Obesity or Overweight with ≥1 Weight-related Comorbid Condition (without Type 2 Diabetes)
Abbreviation: W72 TRE = treatment effect under treatment regimen estimand at Week 72.
Displayed results are from randomized patients and treatment regimen estimand data
points set. (1) Observed mean value from Week 0 to Week 72, and (2) model-based estimate
± standard error at Week 72 using primary modified multiple imputation.
Figure 2: Change Over Time in Body Weight in Trial 2 in Patients with Obesity or Overweight and Type 2 Diabetes
Abbreviation: W72 TRE = treatment effect under treatment regimen estimand at Week 72.
Displayed results are from randomized participants and treatment regimen estimand data points set. (1) Observed mean value from Week 0 to Week 72, and (2) model-based estimate ± standard error at Week 72 using primary modified multiple imputation.
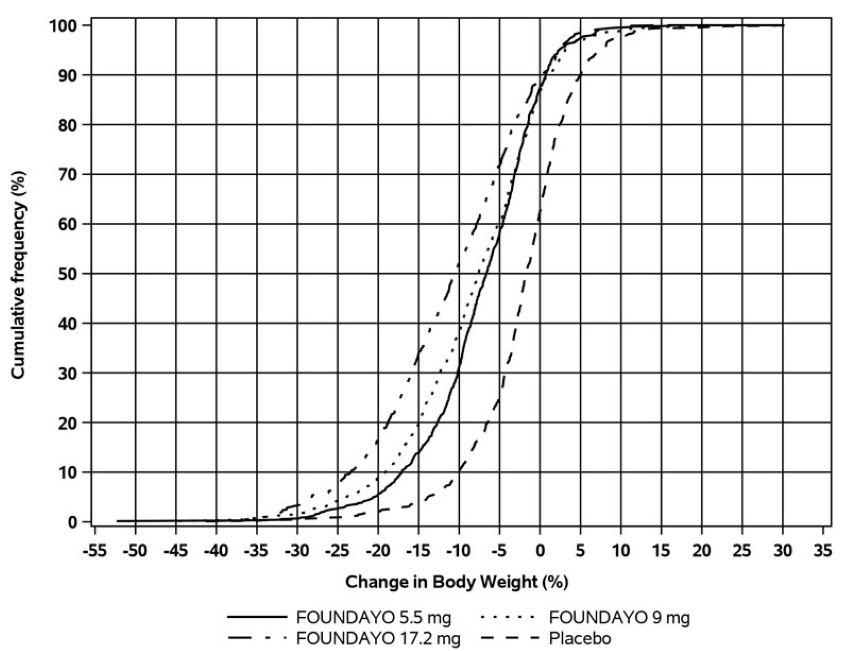
The cumulative frequency distributions of percentage change in body weight are shown
in Figure 3 for Trial 1 and Figure 4 for Trial 2. One way to interpret this figure is to select a change in body weight
of interest on the horizontal axis and note the corresponding proportions of patients
(vertical axis) in each treatment group who achieved at least that degree of weight
reduction. For example, the vertical line arising from -10% in Figure 3 intersects the FOUNDAYO 17.2 mg once daily and placebo curves at approximately 55%
and 13%, respectively, which correspond to the values shown in Table 6.
Figure 3: Changes in Body Weight (%) from Baseline to Week 72 in Trial 1 in Patients with Obesity or Overweight with ≥1 Weight-related Comorbid Condition (without Type 2 Diabetes)
Note: Based on average percent weight change of each randomized patient within each
specific treatment group from 100 imputed datasets including observed data and imputed
data using the primary modified multiple imputation method for missing values.
Figure 4: Changes in Body Weight (%) from Baseline to Week 72 in Trial 2 in Patients
with Obesity or Overweight and Type 2 Diabetes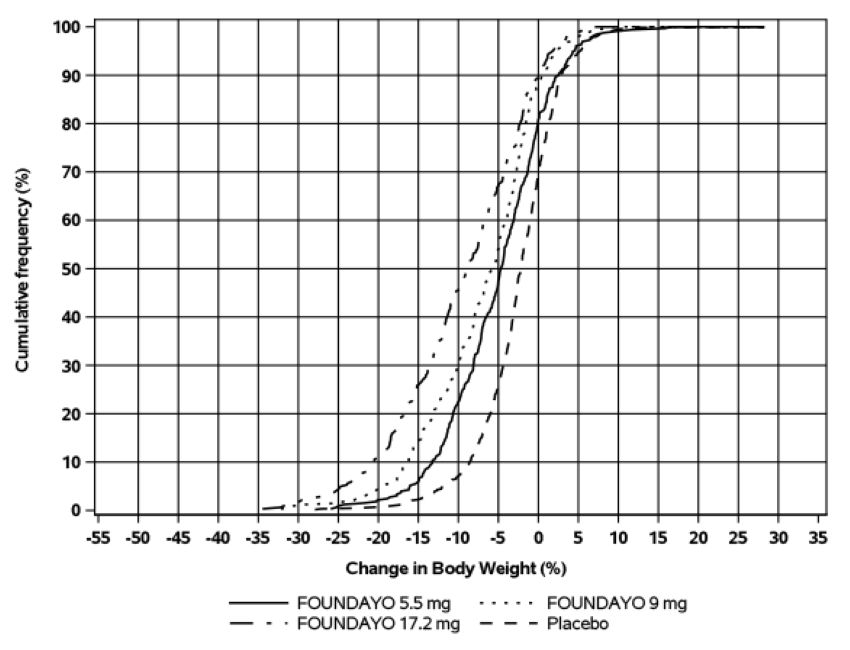
Based on average percent weight change of each randomized patient within each specific treatment group from 100 imputed datasets including observed data and imputed data using the primary modified multiple imputation method for missing values.
Effect of FOUNDAYO on Anthropometry and Cardiometabolic Parameters in Trials 1 and 2
Changes in waist circumference and cardiometabolic parameters with FOUNDAYO are shown in Table 7 for Trial 1 and Trial 2.
|
Abbreviations: ANCOVA = analysis of covariance; CI = confidence interval; N = number of patients randomly assigned to trial drug. Note: Baseline mean is calculated using all randomized patients. For patients with missing baseline values, imputed values are used. |
||||||||
|
a The intent-to-treat population consists of all randomized patients. Missing data were imputed from retrieved patients of the same randomized treatment group when the missingness was possibly related to trial treatment; otherwise, missing data were imputed using observed data from the same randomized treatment group (primary modified multiple imputation). |
||||||||
|
b Unconditional average treatment effect estimated using ANCOVA adjusted for baseline value and other stratification factors. |
||||||||
|
c p<0.001 (unadjusted 2-sided) compared to placebo for superiority; controlled for multiplicity. |
||||||||
|
d Not controlled for multiplicity. |
||||||||
|
e Baseline value is the geometric mean. |
||||||||
|
f Analyzed using log-transformed data. |
||||||||
| Trial 1 (without diabetes) | Trial 2 (with type 2 diabetes) | |||||||
| Intent-to-Treat (ITT) Populationa | Placebo once daily N = 949 |
FOUNDAYO 5.5 mg once daily N = 723 |
FOUNDAYO 9 mg once daily N = 725 |
FOUNDAYO 17.2 mg once daily N = 730 |
Placebo once daily N = 630 |
FOUNDAYO 5.5 mg once daily N = 329 |
FOUNDAYO 9 mg once daily N = 332 |
FOUNDAYO 17.2 mg once daily N = 322 |
| Waist Circumference (cm) | ||||||||
| Baseline mean | 112.8 | 112.2 | 112 | 112.4 | 115 | 116.8 | 116.2 | 114.7 |
| Change from baselineb | -3.1 | -7 | -8.2 | -10 | -2.8 | -5.4 | -6.3 | -8.3 |
| Difference from placebo (95% CI)b | -3.9 (-4.7, -3.1)c |
-5.1 (-5.9, -4.2)c |
-6.9 (-7.8, -6)c |
-2.6 (-3.5, -1.6)d |
-3.5 (-4.4, -2.6)d |
-5.5 (-6.5, -4.6)c |
||
| Systolic Blood Pressure (mm Hg) | ||||||||
| Baseline mean | 125.8 | 125.4 | 125.1 | 125.8 | 130.6 | 131.3 | 132.1 | 132.5 |
| Change from baselineb | -1.4 | -5.7 | -5.1 | -6.4 | -1.5 | -4 | -4.2 | -4.6 |
| Difference from placebo (95% CI)b | -4.2 (-5.4, -3)d |
-3.7 (-4.8, -2.5)d |
-4.9 (-6.1, -3.8)d |
-2.5 (-4.1, -0.8)d |
-2.7 (-4.4, -1)d |
-3 (-4.8, -1.2)d |
||
| Diastolic Blood Pressure (mm Hg) | ||||||||
| Baseline mean | 81.8 | 81 | 81.2 | 80.9 | 81 | 81.6 | 82.1 | 81.8 |
| Change from baselineb | -1.4 | -2.4 | -2.3 | -2.7 | -1.3 | -1.4 | -1.4 | -1.8 |
| Difference from placebo (95% CI)b | -0.9 (-1.7, -0.1)d |
-0.8 (-1.7, 0)d |
-1.2 (-2, -0.4)d |
-0.1 (-1.2, 0.9)d |
-0.1 (-1.2, 0.9)d |
-0.6 (-1.6, 0.5)d |
||
| Pulse Rate (beats per minute) | ||||||||
| Baseline mean | 73.7 | 73 | 73 | 73.5 | 74.2 | 75.9 | 73.7 | 74.6 |
| Change from baselineb | 0.6 | 3.6 | 3.9 | 4.6 | 0.4 | 2.8 | 3.9 | 3.6 |
| Difference from placebo (95% CI)b | 3.1 (2.2, 4)d |
3.4 (2.5, 4.2)d |
4.1 (3.1, 5)d |
2.4 (1.3, 3.6)d |
3.5 (2.4, 4.6)d |
3.2 (2, 4.4)d |
||
| HbA1c (%) | ||||||||
| Baseline mean | 5.6 | 5.6 | 5.6 | 5.6 | 8 | 8 | 8.1 | 8.1 |
| Change from baselineb | -0.1 | -0.3 | -0.3 | -0.3 | -0.4 | -1.2 | -1.4 | -1.7 |
| Difference from placebo (95% CI)b | -0.2 (-0.2, -0.2)d |
-0.2 (-0.2, -0.2)d |
-0.3 (-0.3, -0.2)d |
-0.8 (-1, -0.6)c |
-1 (-1.2, -0.8)c |
-1.2 (-1.4, -1.1)c |
||
| Total Cholesterol (mg/dL) | ||||||||
| Baseline meane | 192.6 | 192.5 | 191.2 | 192.2 | 167.6 | 168 | 167.4 | 167.6 |
| Percent change from baselineb | -1.9 | -3.4 | -4.5 | -4.3 | -2.2 | -1.5 | -2 | -4.9 |
| Relative difference from placebo (95% CI)b | -1.6 (-3.1, 0)d,f |
-2.7 (-4.2, -1.1)d,f |
-2.5 (-4, -0.9)d,f |
0.7 (-2.2, 3.6)d,f |
0.1 (-2.6, 2.9)d,f |
-2.7 (-5.3, -0.1)d,f |
||
| Non-HDL Cholesterol (mg/dL) | ||||||||
| Baseline meane | 142.3 | 141.9 | 139.8 | 142.5 | 122.2 | 120.7 | 121.8 | 121 |
| Percent change from baselineb | -2 | -5.4 | -7 | -7.7 | -3 | -4.3 | -4.7 | -9.7 |
| Relative difference from placebo (95% CI)b | -3.5 (-5.5, -1.4)d,f |
-5.1 (-7, -3.1)d,f |
-5.8 (-7.8, -3.8)d,f |
-1.3 (-5.1, 2.7)d,f |
-1.7 (-5.4, 2.1)d,f |
-6.9 (-10.2, -3.4)d,f |
||
| LDL Cholesterol (mg/dL) | ||||||||
| Baseline meane | 114.4 | 115 | 113.3 | 114.6 | 84.7 | 84.3 | 84.1 | 85.3 |
| Percent change from baselineb | -1.6 | -3.8 | -5.5 | -4.9 | -3.1 | -0.2 | -0.7 | -5.5 |
| Relative difference from placebo (95% CI)b | -2.2 (-4.5, 0.1)d,f |
-4 (-6.2, -1.7)d,f |
-3.3 (-5.5, -1)d,f |
3.1 (-2.2, 8.6)d,f |
2.5 (-2.1, 7.4)d,f |
-2.4 (-6.8, 2.2)d,f |
||
| HDL Cholesterol (mg/dL) | ||||||||
| Baseline meane | 47.8 | 48.1 | 48.6 | 47 | 42 | 43.5 | 42.8 | 43.1 |
| Percent change from baselineb | -1 | 2.1 | 3.1 | 4.5 | 0.8 | 5.8 | 5.3 | 8 |
| Relative difference from placebo (95% CI)b | 3.1 (1.4, 4.8)d,f |
4.1 (2.3, 5.8)d,f |
5.5 (3.8, 7.2)d,f |
5 (2.5, 7.6)d,f |
4.4 (2.1, 6.8)d,f |
7.2 (4.7, 9.7)d,f |
||
| Triglycerides (mg/dL) | ||||||||
| Baseline meane | 125.3 | 121.1 | 119.2 | 125.6 | 162.4 | 157.8 | 164.4 | 157.2 |
| Percent change from baselineb | -4 | -10.4 | -13.1 | -20 | -4.1 | -13.3 | -14.7 | -19.4 |
| Relative difference from placebo (95% CI)b | -6.7 (-9.9, -3.3)d,f |
-9.4 (-12.6, -6.1)d,f |
-16.6 (-19.5, -13.6)d,f |
-9.6 (-14.5, -4.4)d,f |
-11 (-15.6, -6.2)d,f |
-15.9 (-20.3, -11.3)d,f |
||
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
FOUNDAYO (orforglipron) tablets are supplied in bottles with desiccant and child-resistant closure in cartons as follows:
| Tablet Strength of Orforglipron | Description | Package Configuration | NDC Number |
| 0.8 mg | Pink round tablet debossed with “L” on one side and “G1” on the other side | Bottles of 30 tablets | 0002-4178-31 |
| 2.5 mg | Light yellow round tablet debossed with “L” on one side and “G2” on the other side | Bottles of 30 tablets | 0002-4503-31 |
| 5.5 mg | Grayish purple round tablet debossed with “L” on one side and “G3” on the other side | Bottles of 30 tablets | 0002-4794-31 |
| 9 mg | Pink modified oval tablet debossed with “Lilly” on one side and “G4” on the other side | Bottles of 30 tablets | 0002-4803-31 |
| 14.5 mg | Light yellow modified oval tablet debossed with “Lilly” on one side and “G5” on the other side | Bottles of 30 tablets | 0002-4839-31 |
| 17.2 mg | Grayish purple modified oval tablet debossed with “Lilly” on one side and “G6” on the other side | Bottles of 30 tablets | 0002-4953-31 |
16.2 Storage and Handling
- Store FOUNDAYO at room temperature 20°C to 25° C (68°F to 77° F); excursions permitted between 15°C and 30°C (59°F and 86°F) [See USP Controlled Room Temperature].
- FOUNDAYO is light sensitive. Protect from light by keeping in the original bottle and carton and replacing the cap each time after opening. If the carton is no longer available, store bottle away from light.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Risk of Thyroid C-Cell Tumors
Inform patients that in studies with rodents, medicines that work like FOUNDAYO caused thyroid C-cell tumors and that the human relevance of this finding has not been determined. Counsel patients to report symptoms of thyroid tumors (e.g., a lump in the neck, persistent hoarseness, dysphagia, or dyspnea) to their healthcare provider [see Boxed Warning, Warnings and Precautions (5.1)].
Acute Pancreatitis
Inform patients of the potential risk for acute pancreatitis and its symptoms: severe abdominal pain that may radiate to the back, and which may or may not be accompanied by nausea or vomiting. Instruct patients to discontinue FOUNDAYO promptly and contact their healthcare provider if pancreatitis is suspected [see Warnings and Precautions (5.2)].
Severe Gastrointestinal Adverse Reactions
Inform patients of the potential risk of severe gastrointestinal adverse reactions. Instruct patients to contact their healthcare provider if they have severe or persistent gastrointestinal symptoms [see Warnings and Precautions (5.3)].
Acute Kidney Injury Due to Volume Depletion
Inform patients of the potential risk of acute kidney injury due to dehydration and volume depletion associated with gastrointestinal adverse reactions. Advise patients to take precautions to avoid fluid depletion. Inform patients of the signs and symptoms of acute kidney injury and instruct them to promptly report any of these signs or symptoms or persistent (or extended) nausea, vomiting, and diarrhea to their healthcare provider [see Warnings and Precautions (5.4)].
Hypoglycemia
Inform patients of the risk of hypoglycemia and educate patients on the signs and symptoms of hypoglycemia. Advise patients taking insulin or insulin secretagogue therapy such as a sulfonylurea that they may have an increased risk of hypoglycemia when using FOUNDAYO and to report signs and/or symptoms of hypoglycemia to their healthcare provider [see Warnings and Precautions (5.5)].
Hypersensitivity Reactions
Inform patients that serious hypersensitivity reactions have been reported with use of FOUNDAYO. Advise patients on the symptoms of hypersensitivity reactions and instruct them to stop taking FOUNDAYO and seek medical advice promptly if such symptoms occur [see Warnings and Precautions (5.6)].
Diabetic Retinopathy Complications in Patients with Type 2 Diabetes
Inform patients with type 2 diabetes to contact their healthcare provider if they experience changes in vision during treatment with FOUNDAYO [see Warnings and Precautions (5.7)].
Acute Gallbladder Disease
Inform patients of the risk of acute gallbladder disease. Instruct patients to contact their healthcare provider for appropriate clinical follow-up if gallbladder disease is suspected [see Warnings and Precautions (5.8)].
Pulmonary Aspiration During General Anesthesia or Deep Sedation
Inform patients that FOUNDAYO may cause their stomach to empty more slowly which may lead to complications with anesthesia or deep sedation during planned surgeries or procedures. Instruct patients to inform healthcare providers prior to any planned surgeries or procedures if they are taking FOUNDAYO [see Warnings and Precautions (5.9)].
Pregnancy
Advise a pregnant patient of the potential risk to a fetus. Advise patients to inform their healthcare provider if they are pregnant or intend to become pregnant during treatment with FOUNDAYO. Advise women of childbearing potential to use effective contraception during treatment with FOUNDAYO [see Use in Specific Populations (8.1, 8.3)]. Advise patients that there will be a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to FOUNDAYO during pregnancy [see Use in Specific Populations (8.1)].
Contraception
Use of FOUNDAYO may reduce the efficacy of oral hormonal contraceptives. Advise patients using oral hormonal contraceptives to switch to a non-oral contraceptive method or add a barrier method of contraception for 30 days after initiation with FOUNDAYO and for 30 days after each dose escalation [see Drug Interactions (7.3), Use in Specific Populations (8.3), Clinical Pharmacology (12.3)].
Administration
Inform patients to take one FOUNDAYO tablet orally once daily with or without food. Advise patients to swallow tablets whole and not to break, crush, or chew tablets [see Dosage and Administration (2.1)]. Advise patients to keep FOUNDAYO in the original bottle and carton to protect from light [see How Supplied/Storage and Handling (16.2)].
Missed Doses
Inform patients if a dose of FOUNDAYO is missed, it should be taken as soon as possible. Instruct patients not to double up the next dose. If 7 or more consecutive doses are missed, instruct patients to restart dosage escalation at a lower dosage [see Dosage and Administration (2.3)].
Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA
Copyright © 2026, Eli Lilly and Company. All rights reserved.
Pat.: www.lilly.com/patents
OFG-0001-USPI-APRIL2026
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Issued: 04/2026 |
|||||||
|
OFG-0001-MG-APRIL2026 |
||||||||
| Medication Guide FOUNDAYO™ [fown-DAY-oh] (orforglipron) tablets, for oral use |
||||||||
|
What is the most important information I should know about FOUNDAYO?
|
||||||||
What is FOUNDAYO?
|
||||||||
Do not use FOUNDAYO if:
|
||||||||
Before taking FOUNDAYO, tell your healthcare provider about all of your medical conditions,
including if you:
|
||||||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
FOUNDAYO may affect the way some medicines work, and some medicines may affect the
way FOUNDAYO works.
Before using FOUNDAYO, talk to your healthcare provider about low blood sugar and how to manage it. Tell your healthcare provider if you are taking medicines to treat diabetes including an insulin or sulfonylurea. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
||||||||
How should I take FOUNDAYO?
|
||||||||
| What are the possible side effects of FOUNDAYO? FOUNDAYO may cause serious side effects, including:
|
||||||||
|
||||||||
| The most common side effects of FOUNDAYO include: | ||||||||
|
|
|
||||||
| Talk to your healthcare provider if you have any side effect that bothers you or that
does not go away. These are not all the possible side effects of FOUNDAYO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||||||
How should I store FOUNDAYO?
|
||||||||
| Keep FOUNDAYO and all medicines out of the reach of children. | ||||||||
| General information about the safe and effective use of FOUNDAYO. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use FOUNDAYO for a condition for which it was not prescribed. Do not give FOUNDAYO to other people, even if they have the same condition you have. It may harm them. You can ask your pharmacist or healthcare provider for information about FOUNDAYO that is written for health professionals. |
||||||||
| What are the ingredients in FOUNDAYO? Active ingredient: orforglipron Inactive ingredients: Copovidone, crospovidone, magnesium stearate, microcrystalline cellulose, and sodium carbonate anhydrous. The film coating contains polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. The film coating of the 0.8 mg and 9 mg tablets also contains ferric oxide red and ferric oxide yellow, 2.5 mg and 14.5 mg tablets also contains ferric oxide yellow, and 5.5 mg and 17.2 mg tablets also contains ferrosoferric oxide and ferric oxide red. FOUNDAYO™ is a trademark of Eli Lilly and Company. Marketed by: Lilly USA, LLC, Indianapolis, IN 46285, USA Copyright © 2026, Eli Lilly and Company. All rights reserved. For more information, go to www.foundayo.com or call 1-800-545-5979 |
||||||||
PACKAGE CARTON - FOUNDAYO 0.8 mg 30 Count Tablets
NDC 0002-4178-31
Rx only
FOUNDAYOTM
(orforglipron)
tablets
0.8 mg
Dispense with enclosed Medication Guide.
30 tablets
Lilly
PACKAGE CARTON - FOUNDAYO 2.5 mg 30 Count Tablets
NDC 0002-4503-31
Rx only
FOUNDAYOTM
(orforglipron)
tablets
2.5 mg
Dispense with enclosed Medication Guide.
30 tablets
Lilly
PACKAGE CARTON - FOUNDAYO 5.5 mg 30 Count Tablets
NDC 0002-4794-31
Rx only
FOUNDAYOTM
(orforglipron)
tablets
5.5 mg
Dispense with enclosed Medication Guide.
30 tablets
Lilly
PACKAGE CARTON - FOUNDAYO 9 mg 30 Count Tablets
NDC 0002-4803-31
Rx only
FOUNDAYOTM
(orforglipron) tablets
9 mg
Dispense with enclosed Medication Guide.
30 tablets
Lilly